|
This program is very useful for the chemestry student. mirage OS compatible and it cleans up after itself! NOTE: "book value"=accepted value & "Lab value"=experimental value This program is a must-have for chemestry students! it has a pretty neat GUI and is bug-free. Please read the readme file for information on enabling lowercase letters on your calculator. A formula like "CuCl2*5H2O" takes less than a second on a TI-84 Plus), and even accepts hydrates and nested parentheses. I have built that into the program now, but no (ENM's) have yet been released.Įnter the order of the chemical equation, the initial concentration, the constant of the reaction, and find the half-life timeĪ small and very fast program that prompts you for any chemical formula and outputs everything you ever wanted to know about it: its percent composition, molar mass, solubility, and (if it's a hydrocarbon) its complete, balanced combustion reaction equation. This program also will have External Note Modules. This program performs 10 chemistry conversions, and has 15 self contained sub-programs from Modern Chemistry (HOLT, RINEHART and WINSTON), and also has 10 pages of notes (Octet rule, Common Polyatomic Ions, Stock System, Naming Ions, Mass Energy Conversion, Planck's Photon Energy, Ideal Gas Law, Combined Gas Law, Dalton's Partial Pressure, Graham's Law effusion diffusion, Definition of pH, Freezing Point Depression, and Boiling Point Elevation). This program is used to convert pH to to pOH, the Solves for pH of strong and weak acids, buffers,pH during titrations and more.Īcidity is a program that calculates the pH of an acid or base substance. This simple, easy program helps calculate pH, pOH,, and for acid and base problems. Since the size is more than the amount of ram, it automatically archives things for you. Includes common ions with subscripts and superscripts, mole problem help, stoich problem help, molar masses, naming chart, % composition, limiting reactions, electron configurations, chemical equation balancer, and notes. Probably the best chemistry program as of now. Check out the screenshots you'll be amazed. More general features include 326 different unit conversions (in an extremely simple scrollable graphical interface) and many other essential features, such as chemical equation balancing and formula interpreting. Includes 18 different area, volume, and surface area solvers for geometry students, a list of essential trigonometric identities, and many intuitive calculators for many other topics, such as a complete triangle solver. Featuring an easy-to-use menu system, it packs dozens of extremely useful algebra, geometry, calculus, chemistry, and physics functions for middle school, high school and even college courses. This is one of the most complete suites of homework help functions in existence, and it's all packed in one program (with an assembly subprogram for some of the chemistry functions). Português: Lista de iões organizados por carga. Up to TI-83/84 Plus BASIC Science Programs Icon legend: File with screen shots File with animated screen shots File with reviews Featured programs Click a folder name to view files in that folder. TI-83/84 PLUS BASIC SCIENCE PROGRAMS (CHEMISTRY)Ĭlick a filename to download that file. TI-83/84 Plus BASIC Science Programs (Chemistry) So technically, both atomic mass and average atomic mass are atomic masses, but one represents a single atom, and the other represents the average of the isotopes.TI-83/84 Plus BASIC Science Programs (Chemistry). It is the abundance of isotopes of an element found naturally, expressed in percentages. Its unit is also amu.īut the average atomic mass depends on one more critical aspect, the isotopic abundance. The average atomic mass expresses the atomic mass of elements with isotopes. Isotopes became the reason for calculating the average atomic masses, as we must consider an element's isotopes. Then came isotopes, the atoms that differ slightly in atomic masses due to the varying number of neutrons in their nucleus. 
The unit of atomic mass is non-SI, amu (atomic mass unit). This calculation gives us the mass of a single atom of an element.  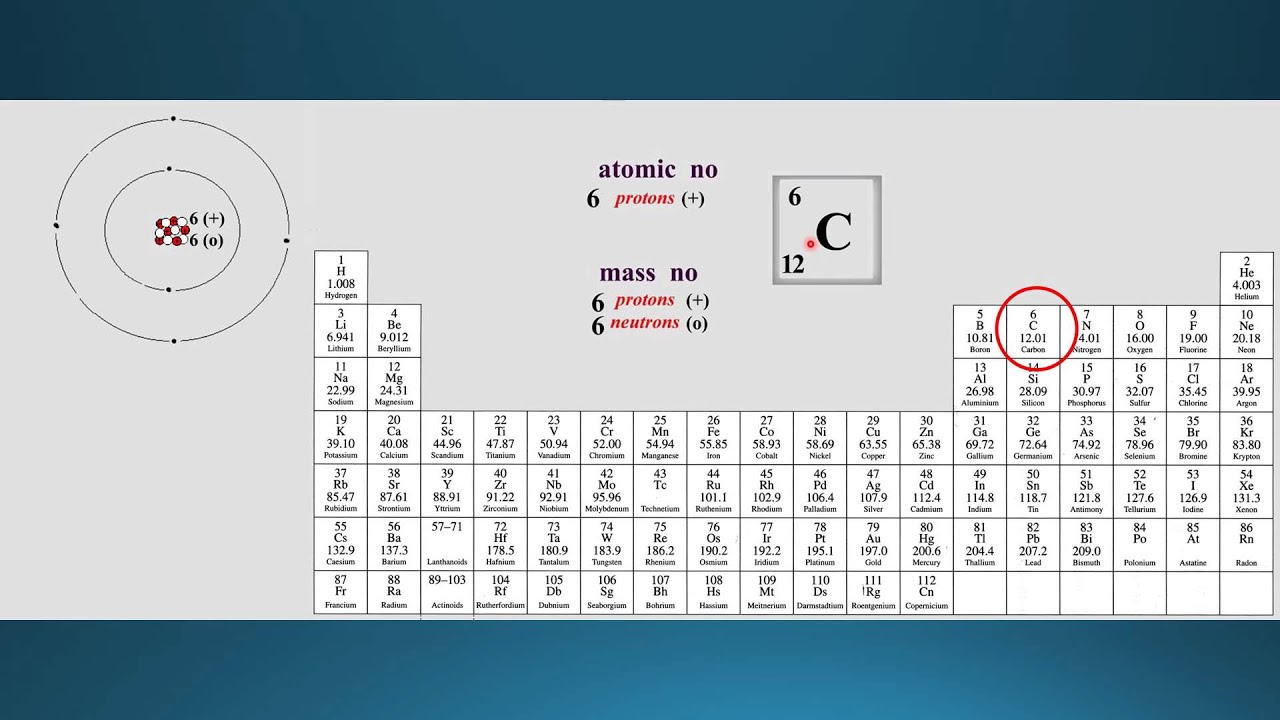
But we can figure it out by adding up the number of protons and neutrons in the nucleus of an atom. It is the mass of a single atom of that element.Įxperimentally it is calculated by mass spectrometry (an analytical technique used to measure the mass-to-charge ratio of ions). We have been familiar with the atomic mass of an element since we started learning about elements and their atoms.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
